Online scheduling is available only to those individuals who have received training on equipment. Review the Bioimaging guideline on how to receive training.


Objectives:
Confocal Specifications:
Mutliphopton Specifications:
Book Now (Room 778)
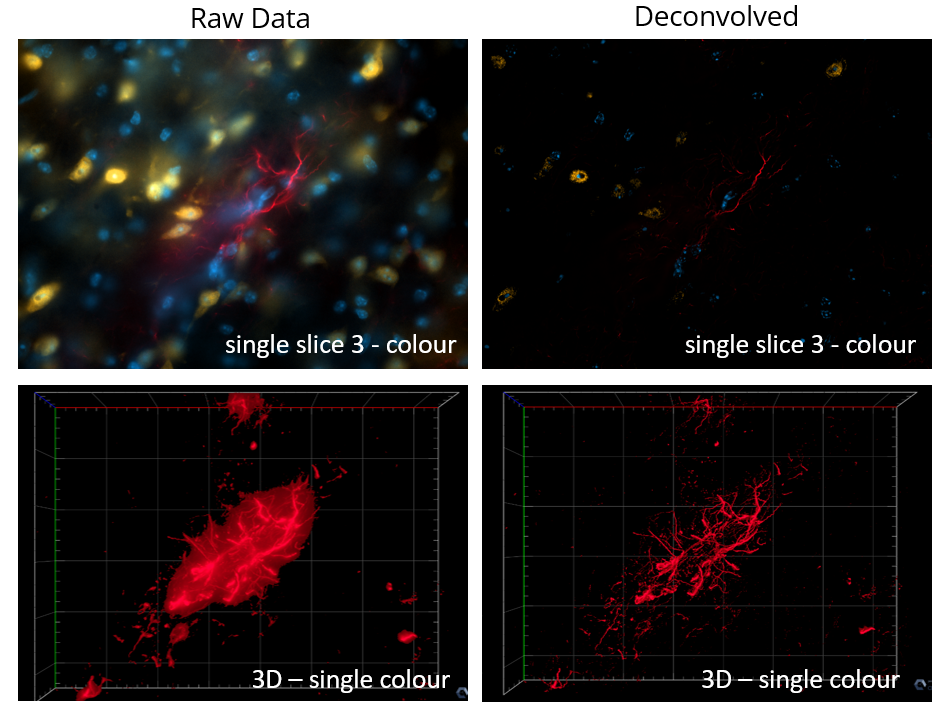




Slide scanning will be offered as a service, please watch video instructions and submit a requisition form.
Objectives:
Brightfield illumination source:
Brightfield Camera – Hitachi 3-Chip color camera, model HV-F202SCL:
Fluorescent Illumination (COLIBRI LED):
Fluorescent emission filters:
Fluorescent Camera- Hamamatsu ORCA-Flash 4.0 V3:

5 HALO modules, two standard modules and three elective modules
The Newton 7.0 FT500 is capable of both bioluminescence (2D and 3D tomography) as well as fluorescence imaging.
Designed for preclinical in vivo imaging of small animals with a fully automated heated stage and an integrated anesthesia system – allowing for up to 5 mice to be imaged simultaneously. (Also capable of ex vivo and in vitro applications.)
Camera – wide aperture (f/0.7) 16-bit cooled CCD camera
Fluorescence – 8 LED excitation channels, and 8 narrow bandpass emission filters for a wide array of fluorophores (420nm – 780nm).
